Article Prepared By: Izwaharyanie Ibrahim
Source: Advanced Functional Materials (Wiley-VCH GmbH)
In the evolving landscape of environmental sensing, the detection of per- and polyfluoroalkyl substances (PFAS) has remained a formidable challenge due to their stability, solubility, and presence at trace concentrations. Instead of designing complex recognition chemistries, researchers are now exploring nature-inspired physicochemical phenomena—and one such breakthrough is the fluorophobic effect.
Zanotti et al. (2025) present a novel approach that exploits the inherent repulsion between fluorinated PFAS molecules and non-fluorous hydrophobic surfaces to create selective electrochemical sensors without requiring PFAS-specific receptors [1].
PFAS: Hidden but Harmful
PFAS are widely used in industrial and consumer products such as Teflon, firefighting foams, and water-repellent coatings [2]. Their strong carbon-fluorine bonds resist degradation, leading to bioaccumulation and long-term health risks, including thyroid disruption and cancer [3]. Regulatory limits are increasingly stringent, with targets in the parts-per-trillion (ppt) range for drinking water [4].
Electrochemical Sensing: A Practical Frontier
Electrochemical sensors offer an attractive route for real-time, on-site PFAS detection due to:
- High sensitivity
- Portability
- Low cost and operational simplicity [5]
However, most existing designs rely on specific molecular recognition like antibodies, aptamers, or imprinted polymers—many of which are not universal or suffer from cross-reactivity in complex water samples [6].
The Fluorophobic Strategy: Rethinking Selectivity
Instead of recognizing PFAS via binding, Zanotti et al. engineer a non-stick, non-fluorous surface (perfluorinated monolayers) that repels PFAS, displacing redox probes in solution and causing measurable current shifts [1].
Key features include:
- No PFAS-specific binding needed
- Selectivity arises from thermodynamic repulsion
- Modular platform applicable to multiple PFAS species [7]
The sensor demonstrates:
- Detection of PFOS and PFOA at 1 ppt in tap water
- Minimal interference from inorganic ions and natural organic matter [8]
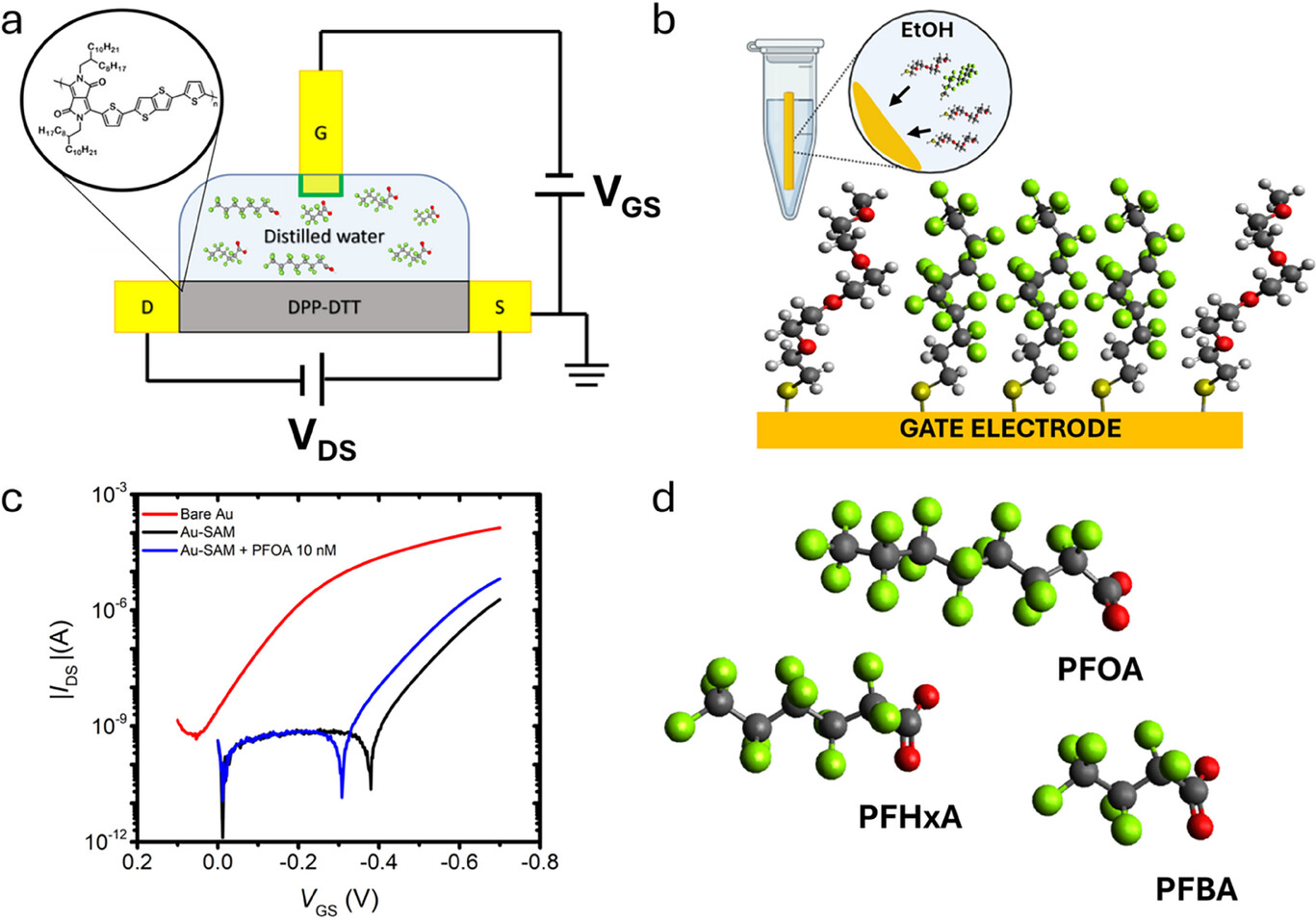
Figure 1- a) Schematic drawing of the PFAS EGOT sensor; inset: structure of the DPP-DTT semiconductor. b) Schematic representation of the mixed SAM. F atoms are depicted in green, O in red, C in gray, S in yellow, and H in white. The drawing on the top left depicts the functionalization of the Au gate electrode incubated in an ethanol binary solution of PFDT and OEG. c) Transfer curves recorded with bare gold (red), SAM-functionalized gold (black) gate electrode, and the latter after exposure to a 10 nm solution of PFOA (blue). d) molecular structures of the three linear PFAS target molecules.
Sensor Design and Working Principle
|
Component
|
Function
|
|
Fluorinated SAM (C8F13–SH)
|
Repels PFAS via fluorophobic interaction [9]
|
|
Methylene Blue (MB) probe
|
Redox signal suppressed when displaced by PFAS [10]
|
|
Gold electrode
|
Provides stable conductive interface [11]
|
When PFAS is introduced, it displaces MB due to its strong fluorophobic interaction, leading to a decrease in electrochemical signal, which is linearly correlated to PFAS concentration [12].
Key Performance Outcomes
- Detection limit: ~1 ppt for PFOS/PFOA in real water
- Response time:
- Selectivity: Outperforms MIP- and aptamer-based systems for mixtures [13]
- Matrix compatibility: Functions in tap water, seawater, and wastewater [14]
- Reusability: Multiple sensing cycles with surface regeneration [15]
Implications for Environmental Monitoring
This method shifts the paradigm from recognition-based to repulsion-based sensing. It offers:
- A universal sensing strategy for multiple PFAS types
- Potential for miniaturization and integration into handheld platforms
- Lower production cost by eliminating biological or synthetic recognition layers [16]
This approach is especially promising for field-deployable systems in resource-limited settings [17].
Perspective from I-AQUAS
I-AQUAS is actively exploring material-driven sensing mechanisms like the fluorophobic effect to enhance monitoring of local water bodies, especially near industrial discharge zones. The simplicity, stability, and adaptability of this approach align with Malaysia’s need for rapid and scalable PFAS detection solutions.
Our next steps include integrating this platform with microfluidic devices and IoT-based data transmission, making environmental sensing smarter and more responsive.
References
- https://advanced.onlinelibrary.wiley.com/doi/full/10.1002/adfm.202508425
- https://www.sciencedirect.com/science/article/pii/S0048969723053895
- https://pubs.acs.org/doi/10.1021/es001834k
- https://pubs.acs.org/doi/full/10.1021/acs.est.1c06896
- https://www.researchgate.net/profile/Hunter-Adams-6
- https://www.sciencedirect.com/science/article/pii/S2214158823000041
- https://pubs.acs.org/doi/10.1021/ac403406d
- https://pubs.acs.org/doi/10.1021/acsami.9b22445
- https://pubs.acs.org/doi/10.1021/jacs.3c03125
- https://pubs.acs.org/doi/10.1021/acs.analchem.6b03522
- https://pubs.acs.org/doi/10.1021/acssensors.6b00481
- https://advanced.onlinelibrary.wiley.com/doi/10.1002/adfm.202313871
- https://advanced.onlinelibrary.wiley.com/doi/10.1002/adma.202410940
- https://pubs.acs.org/doi/10.1021/acsnano.2c08467
- https://pubs.rsc.org/en/content/articlelanding/2009/sm/b813742k
- https://advanced.onlinelibrary.wiley.com/doi/10.1002/aelm.202100071
- https://pubs.acs.org/doi/10.1021/acs.cgd.5b00254
Date of Input: 28/05/2025 | Updated: 11/11/2025 | m_fakhrulddin
MEDIA SHARING